Practical Fragments has
written frequently about pan-assay interference compounds, or PAINS. These molecules
contain substructures that frequently show up in hits that tend not to be
advanceable, often wasting considerable effort. One criticism of the PAINS
concept is that the original definitions were based on a limited number of
screens in one assay format. In a new (open-access) J. Med. Chem. paper, Alison Axtman
and collaborators at University of North Carolina Chapel Hill, Emory University,
and Oxford University characterize one class of PAINS in more detail.
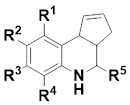
The researchers focused on fused
tetrahydroquinolines, or THQs. Of the 51 molecules containing this substructure
in the original 2010 PAINS paper, 34 hit in at least one of the assays, and one
hit all six. At the time Jonathan Baell and Georgina Holloway noted that “it is
not clear for some PAINS, such as the fused tetrahydroquinolines, what the
relevant mechanisms of interference may be.”
The new paper notes that fused THQs are
common in screening libraries, with more than 15,000 commercially available. They
also frequently show up as hits: the researchers summarize more than two dozen
examples against a wide variety of targets including phosphatases, kinases,
protein-protein-interactions, and more. In most cases the hits are modestly
active, with low to mid micromolar IC50 values, though a few are high
nanomolar.
Promiscuity per se is not
necessarily bad. Just last week we noted that the 7-azaindole fragment was the
starting point for three approved drugs. However, despite showing up as hits in
so many screens, only one peer-reviewed paper reports a crystal structure of a fused
THQ bound to a protein, and the researchers note that “no optimized chemical probes or approved drugs contain the chemotype.”
Importantly, fused THQs hit in a
variety of different assay formats, including spectrophotometric,
chemiluminescent, SPR, and radiochemical. Thus, these are not merely
problematic in the AlphaScreen format studied in the original PAINS paper.
So what’s going on? The researchers
found that, while molecules containing the fused THQ core were initially colorless,
they darkened when dissolved in DMSO or chloroform, turning purple within 72
hours. Interestingly, the reaction seems to be light-dependent: solutions
stored in the dark remained colorless. Thin layer chromatography and NMR showed
new species forming, and mass spectrometry revealed oxidation with loss of two
or four hydrogen atoms. The isolated double bond in the cyclopentene ring seemed
to be the culprit, as the saturated analog was stable. Indeed, all of the hits
shown in the paper contain the double bond, so fused THQs that lack this feature may be
fine – if they ever show up in your assay.
It is still not clear exactly how
the decomposition products light up so many assays, but in general it’s a good
idea to steer clear of molecules that fall apart in ambient light, unless you’re
trying to make a photosensitizer.
The researchers conclude that “it
is tragic to continue to watch groups invest time and resources in dead-end
hit-to-lead campaigns, and the medicinal chemistry community will benefit
everyone if the cycle stops.”
This concludes our public service
announcement.