The phosphatidylinositol-3 kinases (PI3Ks) comprise a family of lipid kinases that are important in a variety of biological pathways and have thus become popular targets for drug-discovery; earlier this year we highlighted a fragment screen out of AstraZeneca against four members of the family. In the most recent issue of Bioorg. Med. Chem. Lett., researchers from Pfizer have published their approach to one of these targets.
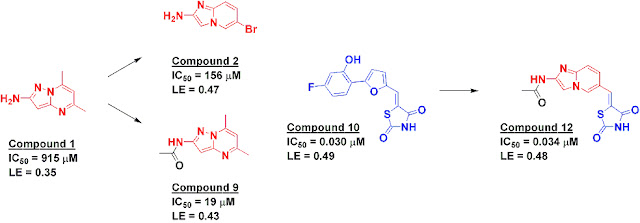
Samantha Hughes and colleagues first tested 5960 fragments in a biochemical assay (at 0.5-1.5 mM) to find molecules that inhibited PI3gamma. Hundreds of hits resulted, of which 150 were confirmed in full dose-response curves. These were further triaged using orthogonal methods, ultimately resulting in five X-ray structures of co-complexes, including compound 1, which binds to the hinge region of the kinase. Virtual screening of the larger Pfizer library led to the discovery of additional compounds such as compound 2. Growing compound 1 by adding an acetyl group generated compound 9, improving both potency and ligand efficiency, but synthetic challenges stymied further work. However, a closer examination of the crystal structure of compound 1 suggested a merging strategy with the previously reported compound 10 to generate compound 12, with high potency and ligand efficiency.
Astute (or paranoid) readers will notice that compound 12 contains a Michael acceptor that looks suspiciously reactive. In fact, it is closely related to the notorious rhodanines, many of which form covalent bonds with proteins and, because of the resulting promiscuity, have been accused of “polluting the literature.” Nonetheless, crystallography revealed that the compound binds (noncovalently) to PI3gamma exactly as designed. Moreover, it is fairly selective for other kinases, inhibiting only 3 out of 43 tested at 1 micromolar compound. Compound 12 is also metabolically stable, permeable, and cell active. This is a good example of why it is important not to be overly dogmatic in compound triaging, particularly at an early stage in a project.
Finally, it is worth noting that this paper comes from the storied Sandwich Laboratories, which are being closed down. If there is a silver lining to this tragedy it is that the closure has resulted in a flurry of nice publications from the site. Still, such papers hardly offset the opportunity cost of the drugs that would otherwise have been discovered there – nor the disruption caused to hundreds of scientists. Practical Fragments wishes the best of luck to all of them.



2 comments:
ok, the rhodanine doesn't covalently bind to the target here, but what about all the other targets it could bind to and related tox issues...rhodhanines why bother?...
I'd guess that the imides in compounds 10 and 12 will be sufficiently acidic to deprotonate under assay conditions and if so this is likely to reduce their electrophilicity in the Michaeil addition. From what I remember TZDs (SMILES for prototype: O=C1NC(=O)SC1 ) typically have a pKa of aboout 6.5).
Post a Comment